AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Compton effect recoil angle of electron11/21/2023 (1), it is seen that the energy would also be decreased. Compton realized though, that if the interaction was modeled as a collision between two particles (electron and photon), the scattered x-rays would-be of longer wave length (compared to the incident-rays) because the recoiling electron would acquire some of the energy and momentum of the incoming x-ray.4 Since wavelength is inversely proportional to frequency, the frequency of the scattered x-rays was less. In other words, the scattered x-rays were of longer wavelength.4 This was contrary to the predictions of classical physics. 1 However, it was observed that x-rays were more easily absorbed by materials than waves of longer wavelength. Hence, the wavelength of radiation from the oscillating electrons would be identical to the wavelength of the incoming xrays. Classically, x-rays would shake the electrons of a target material at the same frequency of the x-ray. Compton theorized and then experimentally demonstrated that electromagnetic waves had the properties of particles. Here I am stuck since this does not give me the required expression for the shift - just transforming frequencies to the corresponding wavelength does not give me the required result.En = nhf (1) where En is the energy, n is a non-negative integer, h is Planck’s constant, and f is the frequency of the photon.2 In 1905, Albert Einstein extended Planck’s inference to include not only black body radiation but all electromagnetic waves! Therefore, Einstein hypothesized that light is quantized with energy proportional to its frequency.3 The obvious principle to be deduced from these discoveries is that light possessed attributes of waves and particles! In 1922, Arthur Holly Compton solidified Planck’s assumption and therefore firmly established a new era of physics. Which after a little bit of algebra becomes $-$ Therefore from equations $(1)$ and $(2)$, we get $-$ 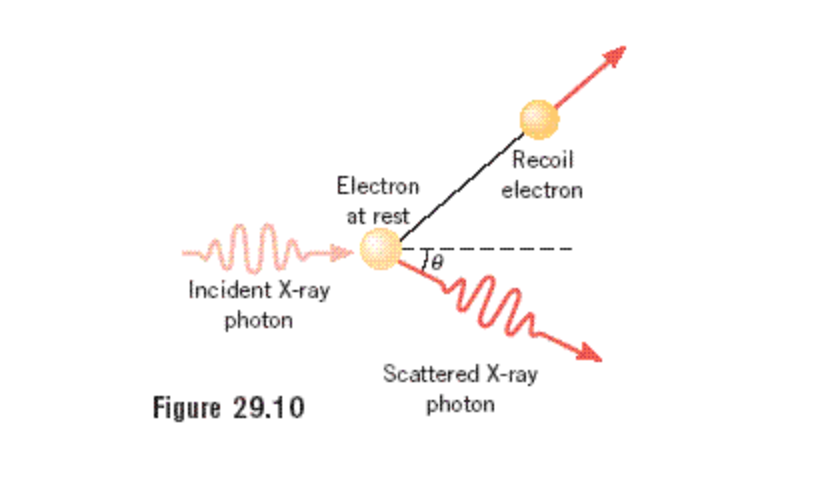
Where $T$ is the energy given to the electron by the photon, which is essentially $h (\nu - \nu_0)$. Now we come to energy conservation: The total energy of the electron is given by $$p c \cos \phi = h \nu_0 + P c - h \nu \cos \theta$$ $$p c \sin \phi = h \nu \sin \theta$$ Where $\nu$ is the frequency of the electron after scattering, $p$ is the momentum of the electron after scattering, and $\phi$ is the electron scattering angle. Then momentum conservation gives, for the $x$- and $y$- axes respectively $-$ Suppose the photon and the electron are both moving initially along the $x$-axis. In proving this, I started in the same way as in the derivation for "stationary electron" - conservation of momentum and energy along each axis. $$\Delta \lambda = 2 \lambda_0 \frac$ is the initial energy of the electron. 
The book tells us that this shift is given by In a problem from Bransden and Joachain's Quantum Mechanics, it is asked to calculate the Compton wavelength shift, but the electron is now moving, with a momentum $P$, in the same direction as the approaching photon.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
